How to turn the wind into liquid fuels

The winds of change are blowing through the energy industry as scientists and engineers seek ever cleaner and more efficient ways to generate power. These same winds can help create clean synthetic alternatives to gasoline that work with existing internal combustion engines, or which can be used in other sectors like the chemical industry.
So how does it work?
Surplus renewable power can be used to drive the electrolysis process in order to create hydrogen. And that hydrogen is then used to help create methanol or other derivatives. The methanol can be either mixed with gasoline or used in place of traditional fuels and chemicals.
The green credentials of synthetic fuels like methanol have attracted attention from countries keen to cut CO2 emission levels. Since 2000, the global methanol market has experienced strong and sustained growth and is set to top more than 11% compound annual growth to 2026, due in part to surging demand from the Asia-Pacific region.
Fuelling the future
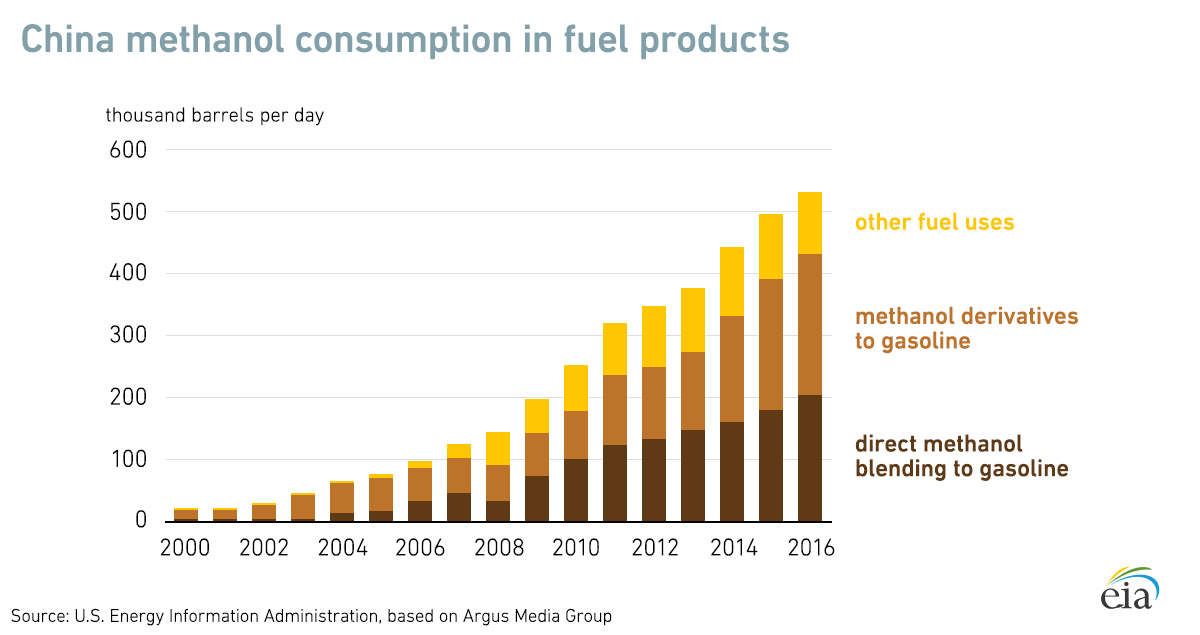
China is the world’s biggest consumer of methanol and the country’s use of liquid fuels has increased sharply over the past two decades.
Other key markets for methanol in the region include Japan and South Korea. And globally, methanol demand has increased by more than 6% in the last five years.
It is the CO2-busting benefits of synthetic fuels that help drive global demand. In China’s transport sector, methanol is commonly used to reduce emissions in rural areas and recently 10,000 methanol propelled vehicles took to the nation’s roads.
Europe saw the introduction of the world’s first methanol-powered ferry, which links the North Sea ports of Kiel and Göteborg.
But there are also other benefits. Synthetic fuels can be easily stored and require little infrastructure investment to bring to market, for example.

Environmental credentials
The creation of liquid fuel alternatives is also a way of exploiting surplus renewable energy which would otherwise be wasted.
On very windy days, surplus energy may be produced which cannot be absorbed by the grid. In mature renewable energy markets like Europe and California, we are already experiencing what we call “curtailing” at work, which means cutting off surplus renewable power generation.
But power-to-fuel technologies means that excess energy from renewables can be stored as synthetic fuel. And as the renewable energy market grows, demand for these technologies will increase.
Decarbonizing the transport sector is not as simple as switching to electric vehicles. Shipping, aviation and land transport cannot be electrified overnight.
And finding other ways to reduce carbon emissions during this transition period will also be essential when it comes to protecting the environment. Down to business
So, we have established there is a market for synthetic fuels. And the legislation is in place to produce them. The question on everyone’s lips now is, how much does it cost?
Producing methanol using carbon-free hydrogen from renewable energy sources is the cleanest way to synthesize fuels, but it is expensive.
Of course, there are less costly ways to produce hydrogen but these are not as clean.
At the moment, price is a challenge that needs to be addressed. But over time I see the gap narrowing. The task ahead involves bridging the price-gap between conventional fuels and their synthetic counterparts.
For methanol produced using electrolysis, the price of synthetic fuel is determined by the cost of the wind power or energy source used to generate the electricity. So, in this case the cheaper the electricity, the greater chance of developing a competitively priced alternative fuel.
Falling costs of renewables are being met with increased pressure for the energy industry to decarbonize, which should increase the viability of producing synthetic fuels over timeMitsubishi Hitachi Power Systems (MHPS) is currently conducting several real-world projects to develop synthetic fuels. The first involves synthesizing dimethyl ether (DME) through electrolysis, for the automobile market. DME is a clean-burning, non-toxic alternative fuel to diesel. We are also working with a German utility company to use hydrogen created through electrolysis to produce liquid methanol.
Projects like these still have some way to go and are not without their challenges. But they could also potentially transform tomorrow’s energy sector, by safeguarding the environment, providing energy storage and offering alternatives to fossil fuels.





